
Most people know the name of a medicine, but not the part that actually makes it work. That part is the active pharmaceutical ingredient (API), and in pharmaceutical manufacturing, its quality is everything.
That is why the ICH Q7 guidelines matter. Developed by the International Council for Harmonisation (ICH), they define the Good Manufacturing Practice (GMP) requirements for API manufacturing. For companies producing or sourcing APIs, the guideline forms the foundation for building quality into the product from starting materials through final release.
In this guide, we’ll explain what ICH Q7 guidance is, what it covers, how it differs from ICH Q7A, and how quality teams translate its principles into reliable API manufacturing practices.
Key takeaways
Why API quality matters: the heparin crisis
In 2008, the pharmaceutical industry was reminded of a hard truth: when API quality fails, the consequences can reach patients fast.
The crisis centered on heparin sodium, the active pharmaceutical ingredient in a widely used anticoagulant medicine. In January of that year, alarm bells began ringing across the United States as hospitals reported severe allergic reactions and dangerously low blood pressure associated with Baxter’s heparin products. Within months, the number of deaths linked to heparin had risen into the dozens, eventually surpassing eighty. Baxter ultimately recalled all of its heparin vials.
Scientists eventually identified an unexpected contaminant: over-sulfated chondroitin sulfate, a cheaper compound derived from animal cartilage that closely resembled heparin and had slipped past routine quality controls.
Investigators traced the contaminant through a complex supply chain to Changzhou SPL, a Chinese supplier of heparin API to Baxter. As the supply network was untangled, regulators discovered that the genuine ingredient had been replaced upstream with the cheaper substitute by suppliers that included unregulated labs and farms.
This is exactly why the ICH Q7 guidelines exist: to provide the quality systems, oversight, and manufacturing discipline required to prevent this type of failure in API production.
Complying with ICH Q7 requires strong control over API manufacturing processes, documentation, and quality activities. Scilife helps pharma teams simplify that complexity with a connected platform built for traceability, consistency, and inspection readiness.
What is ICH Q7?
The International Council for Harmonisation (ICH) brings together regulators from the United States, Europe, and Japan to align technical expectations for pharmaceutical quality, safety, and clinical studies.
One of its quality guidelines is ICH Q7, which focuses specifically on Good Manufacturing Practice for active pharmaceutical ingredients.
In practice, ICH Q7 forms the backbone of API GMP compliance for pharmaceutical companies, biotechnology manufacturers, and contract development and manufacturing organizations (CDMOs).
Scope of ICH Q7 guidelines
ICH Q7 focuses specifically on the manufacture and control of the API, not the finished dosage form.
The ICH Q7 guidelines cover APIs produced through chemical synthesis, extraction, cell culture, or fermentation, recovery from natural sources, or combinations of these approaches.
ICH Q7 guideline for API manufacturing applies from the API starting material, and GMP expectations increase as the process moves through intermediates, purification, physical processing, packaging, storage, and distribution.
What is the difference between ICH Q7 and ICH Q7A?
The guideline was originally issued as ICH Q7A, titled Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients.
Over time, the “A” was dropped in common usage, and today many professionals simply refer to it as ICH Q7. In practice, both terms refer to the same GMP guideline, which is why both names still appear in audits, procedures, and regulatory discussions.
Core GMP areas covered by the ICH Q7 guidelines
ICH Q7 describes a comprehensive operating model for controlling API manufacturing and quality.
It covers areas including:
- Quality management – the structure of the quality system, independence of the quality unit, internal audits, and product quality review.
- Personnel – training, qualifications, hygiene, and responsibilities.
- Buildings and facilities – layout, contamination prevention, utilities, water systems, and sanitation.
- Process equipment – design, maintenance, cleaning, calibration, and computerized systems.
- Documentation and records – control of procedures, manufacturing instructions, laboratory data, and batch records.
- Materials management – receipt, quarantine, sampling, testing, storage, and re-evaluation of materials.
- Production and in-process controls – execution of manufacturing steps and monitoring of process parameters.
- Packaging and identification labelling – ensuring intermediates and APIs are correctly identified and labelled.
- Storage and distribution – protecting API quality during warehousing and transport.
- Laboratory controls – specifications, analytical methods, testing, certificates of analysis, stability programs, and retention samples.
- Validation – demonstrating that processes, cleaning procedures, systems, and analytical methods perform as intended.
- Change control – evaluating and approving changes that may affect product quality.
- Rejection and reuse of materials – handling rejected, reprocessed, reworked, recovered, or returned materials.
- Complaints and recalls – procedures for investigation, trend analysis, and recall decisions.
- Contract manufacturers – oversight and auditing of outsourced operations.
- Agents, brokers, traders, distributors, repackers, and relabellers – quality expectations across the distribution chain.
- Additional guidance for APIs produced by cell culture or fermentation.
- Adapted GMP expectations for APIs used in clinical trials.
Taken together, these areas show that ICH Q7 guidance is not simply a manufacturing rulebook. It is a framework for controlling API quality across operations, laboratories, documentation systems, and supply chains.
Responsibilities of quality teams under ICH Q7
Under ICH Q7, quality is not something that gets checked only at the end of the process. It has to be built into the system that governs how the API is manufactured, tested, reviewed, released, stored, and distributed.
That is why the quality unit plays such a central role. Its responsibility goes far beyond reviewing paperwork or approving the final batch release. The quality unit helps ensure the entire quality system operates as intended — reviewing critical records, investigating deviations, assessing validation activities, approving specifications, overseeing outsourced activities, and ensuring that decisions affecting API quality are documented and traceable.
This broader perspective can be easy to miss when working close to the operational details. One deviation investigation, one supplier qualification, one batch record comment, or one training record review may seem small on its own. But together, these activities provide the evidence that the API remains in a state of control.
That is the core objective of QA under GMP ICH Q7: ensuring the organization can trust its own release decisions. Not because documentation looks complete, but because the manufacturing process, quality systems, and oversight mechanisms collectively demonstrate that the API was consistently produced according to the required standards of quality and purity.
How ICH Q7 fits into a modern pharmaceutical quality system
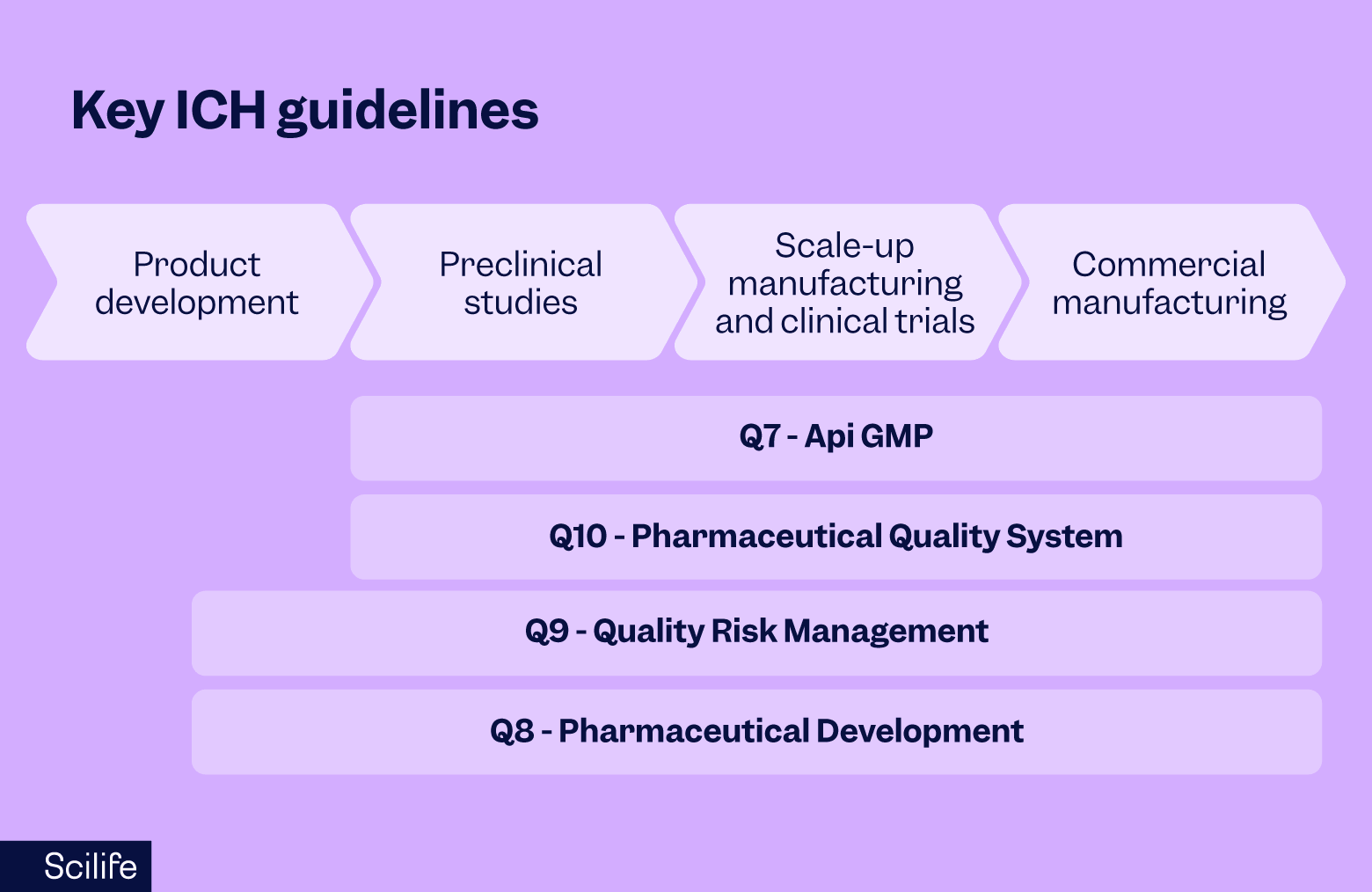
Figure 1. Relationship between key ICH quality guidelines and the lifecycle of a new medicinal product.
At the top of the diagram, the product progresses through four broad stages: product development, preclinical studies, scale-up manufacturing and clinical trials, and commercial manufacturing.
Within this lifecycle, different ICH quality guidelines begin to apply at different stages.
-
ICH Q8 focuses on Pharmaceutical Development. The guideline reflects the principles of Quality by Design (QbD), where the product and manufacturing process are deliberately designed to meet predefined quality requirements. During development, scientists identify critical material attributes and process parameters that influence product quality.
Q8 remains relevant well into commercial manufacturing because the knowledge gained during the posterior stages continues to inform process understanding and improvement.
- ICH Q9, which addresses Quality Risk Management, typically becomes relevant during the middle stages of product development. At that point, companies begin to apply formal risk-based approaches to evaluate which process parameters matter most, where the greatest product risks may exist, and what level of control is needed to protect product quality.
- ICH Q7, which defines GMP requirements for API manufacturing, becomes relevant starting at the preclinical stage. At this point, APIs must be manufactured under sufficient control to support studies such as stability testing and animal studies. As development progresses into clinical manufacturing and commercial production, the expectations defined in ICH Q7 become increasingly formal and comprehensive.
- ICH Q10, the Pharmaceutical Quality System, also begins to take shape during preclinical development. As organizations move toward clinical manufacturing, they require structured systems to manage the quality of the product.
Together, these guidelines form the quality architecture that supports the entire lifecycle of a medicine, from early development through commercial manufacturing.
Implementing ICH Q7 in practice: common API-specific challenges
Although the ICH Q7 guidelines provide a clear regulatory framework, implementation can still be challenging. Many of the most difficult problems in API manufacturing may differ from those seen in finished dosage forms.
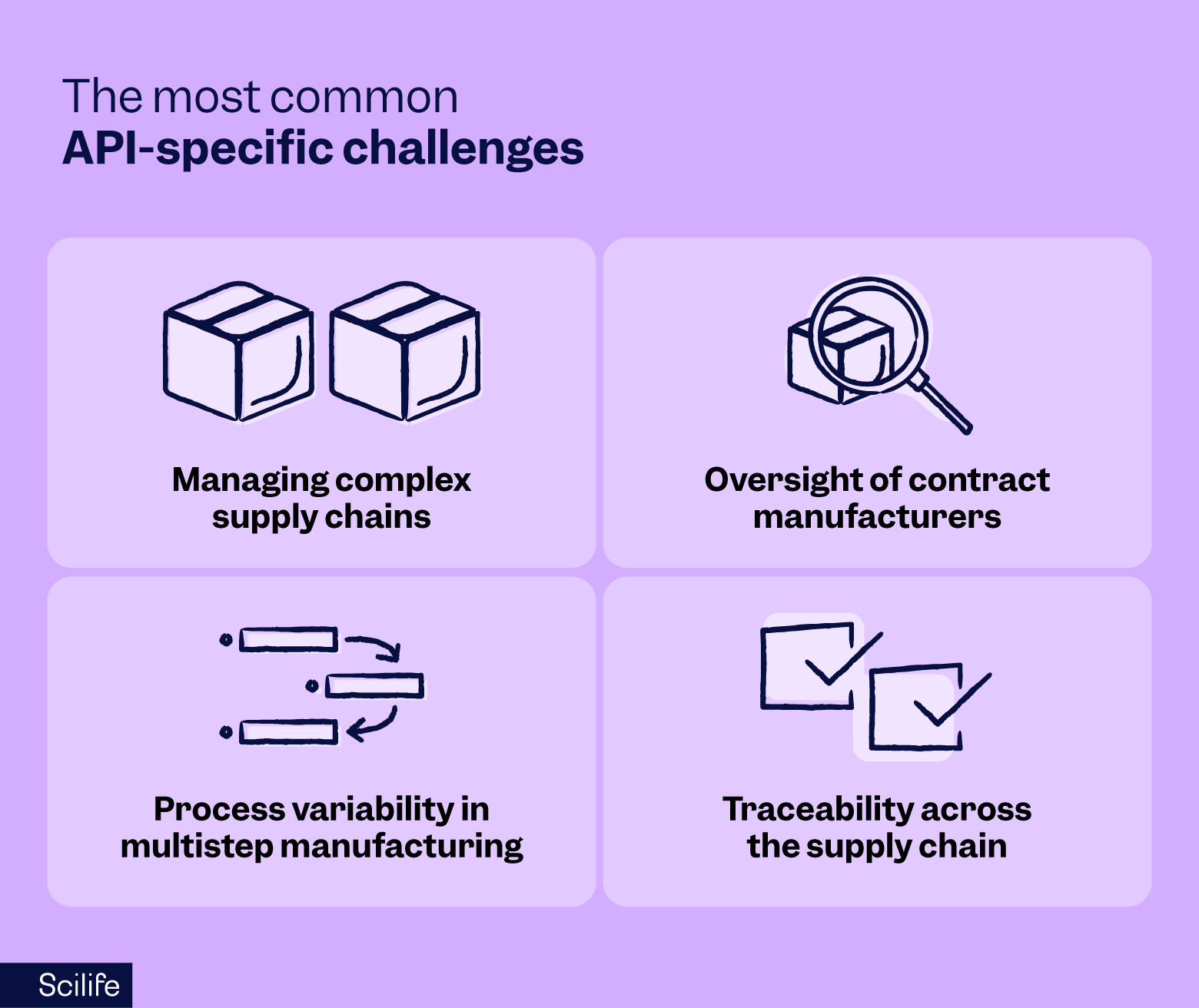
Managing complex supply chains
API manufacturing often relies on networks of suppliers providing starting materials, intermediates, solvents, reagents, and specialized services. When these suppliers operate across multiple regions or countries, maintaining visibility and consistent control becomes significantly more complex.
Oversight of contract manufacturers
Outsourcing is common in API production. Pharmaceutical companies frequently rely on contract manufacturers for intermediates, analytical testing, or even full API manufacturing. Maintaining appropriate oversight, therefore, requires quality agreements, audits, communication channels, and shared documentation systems.
Process variability in multistep manufacturing
API processes often involve complex multistep operations such as synthesis, fermentation, purification, drying, or milling. Small variations in process parameters can influence impurity profiles, yield, or downstream processing performance.
Traceability across the supply chain
Traceability must function in both directions — back to starting materials and suppliers, and forward to the released batches distributed to customers. When deviations, complaints, or recalls occur, this traceability becomes critical for identifying root causes and protecting patients.
Recommended learning:
Compliance tips for supply chain management in the pharma industry.
Conclusion
The ICH Q7 guidelines remain the foundation of GMP compliance for active pharmaceutical ingredients because they connect the daily details of manufacturing, testing, documentation, supplier control, and oversight into a system designed to protect API quality.
In modern pharmaceutical manufacturing, this challenge has only grown. API production is increasingly global, technically complex, and often outsourced across multiple organizations. Managing these systems effectively requires more than procedures alone — it requires visibility, traceability, and quality systems capable of connecting people, records, suppliers, and decisions.
Digital quality platforms can support this work in practical ways. When quality teams need to manage supplier qualification, contract manufacturer documentation, change records, and controlled procedures across multiple partners, connecting those workflows within a unified system becomes a clear operational advantage.
FAQ
What are the main requirements of the ICH Q7 guidelines for active pharmaceutical ingredients?
The ICH Q7 guidelines establish GMP requirements for API manufacturing, covering quality management, facilities and personnel, materials and production controls, laboratory testing and validation, and documentation. They also address supply chain oversight, including supplier qualification, contract manufacturing, change control, and procedures for handling complaints or recalls.
How do I ensure compliance with ICH Q7A during manufacturing?
Compliance with ICH Q7A during manufacturing is achieved by implementing a robust GMP quality system that controls materials, manufacturing processes, documentation, and testing. This includes qualified personnel, validated processes, proper documentation, supplier oversight, and effective systems for deviations, change control, and product release.
What does the ICH Q7 guidance not apply to?
ICH Q7 guidance applies specifically to active pharmaceutical ingredients rather than finished dosage forms. Its focus is the manufacture and control of the API and related operations.
What is the difference between ICH Q7 and 21 CFR Part 211?
ICH Q7 focuses on GMP for active pharmaceutical ingredients, while 21 CFR Part 211 applies to finished pharmaceutical drug products in the United States. The two frameworks are related, but they address different parts of the pharmaceutical manufacturing chain.








