
If you have spent any time on a manufacturing floor, you know the feeling that kicks in when you hear an inspector has just signed into the lobby. I remember my first major site audit vividly. I had spent three weeks checking every single machine maintenance log, only to have the inspector spend hours asking about the directional flow of personnel in the changing rooms.
It was a humbling lesson. How to obtain GMP certification is about proving you have a controlled, predictable, and transparent system.
If you are a life sciences professional in the EU and the US, staying compliant is the only way to keep your doors open. Whether you are aiming for EU GMP certification to access the European Economic Area or preparing for an FDA inspection in the United States, the goal is the same. Making sure that every dose is safe and effective.
In this blog, we will explain why this process is your most valuable business asset. We will break down the regional differences in how to obtain GMP certification, and provide a roadmap to help you move from a state of audit anxiety to a state of constant readiness.
Key takeaways
What is the GMP certification?
GMP certification is a formal verification by a regulatory body, like the EMA in Europe or a third-party auditor in the US, that your facility follows Good Manufacturing Practice standards.
So, how to obtain GMP certification? The process of proving absolute GMP compliance. It is a commitment to the 5 P's. People, Process, Products, Premises, and Procedures. Regulators want to see that your system is designed to prevent errors and contamination every single time.
Recommended learning:
How to obtain GMP certification?
The journey to becoming certified requires meticulous planning. While the technical standards are harmonized globally, the administrative procedures depend on your target market.
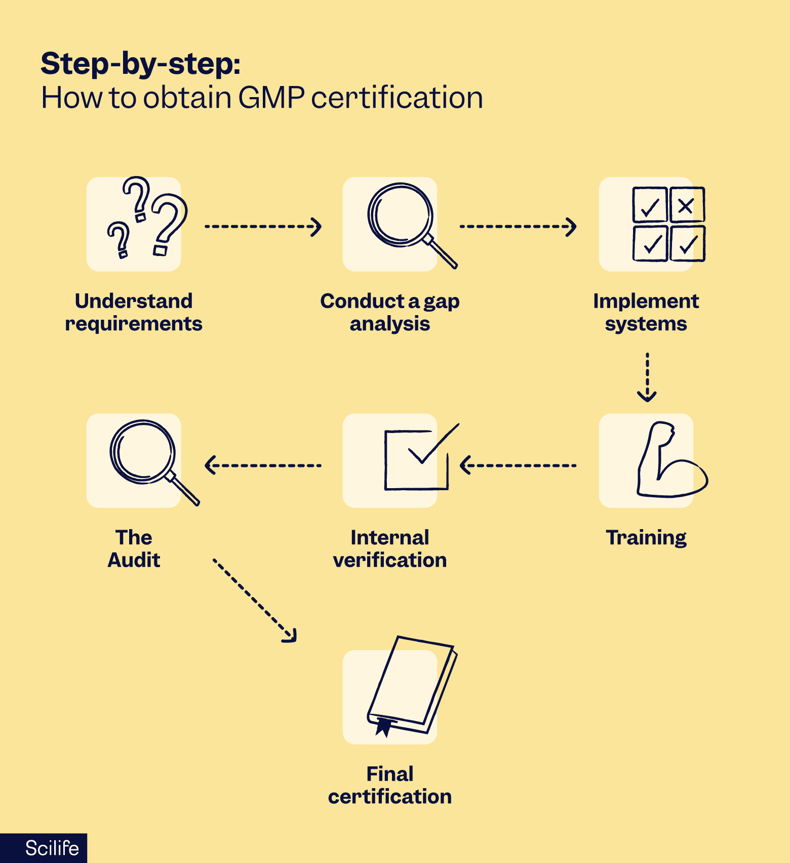
Regardless of whether you are in Paris or Philadelphia, the internal roadmap for how to obtain GMP certification follows a proven seven-step logic:
- Understand requirements: Deep dive into the specific regulations for your product, such as
Annex 1 for steriles or 21 CFR 111 for supplements. - Conduct a gap analysis: This is a search for non-compliance where you must be honest about where your current processes fall short of the standard.
- Implement systems: Fix the gaps by establishing your QMS, securing equipment calibration, and moving toward validated computer systems.
- Training: You can have the best SOPs in the world, but if your personnel are not trained to follow them, you will fail.
- Internal verification: Conduct mock audits to confirm that if you need a training record during a real inspection, you can produce it in minutes.
- The audit: Undergo the official inspection and treat the auditor as a partner in quality rather than an adversary.
- Final certification: Resolve any deficiencies and receive your formal GMP certification.
To understand how to obtain GMP certification for the two most influential regulatory regions, we must look at how authorities verify that products are of consistent high quality and appropriate for their intended use.
How to get an EU GMP certificate?
When manufacturers are looking to distribute in Europe, EU GMP compliance certifications are a legal necessity. The process is coordinated by the European Medicines Agency (EMA) but executed by National Competent Authorities (NCAs), such as the HPRA in Ireland or the ANSM in France.
You cannot get a GMP certificate without first holding a manufacturing or import authorisation from your local NCA. Once you are ready, your site is inspected, usually as part of a marketing authorisation application or a routine risk-based cycle. Inspectors will evaluate your Medical Device File, checking everything from air filtration to your CAPA (Corrective and Preventive Action) logs. If you pass, the NCA issues a formal EU GMP certification and enters it into the public EudraGMDP database. This certificate is typically valid for three years.
How do I become FDA GMP certified in the US?
The US approach is often a point of confusion for EU-based companies. In the United States, the FDA does not issue a standard paper FDA GMP certification for domestic manufacturers.
Instead, they conduct inspections to verify compliance with 21 CFR Parts 210 and 211. You must register your establishment and list your products with the FDA, which then conducts a Pre-Approval Inspection (PAI) for new drugs or a routine surveillance inspection.
If the inspectors find issues, they issue a Form 483. If you address these issues, or if none are found, the FDA issues an Establishment Inspection Report (EIR).
A clean EIR is your proof of compliance. Because the FDA doesn't issue a physical certificate, many US companies hire authorized third parties to conduct a GMP facility certification audit. These third-party certificates are highly valued by business partners and consumers as a sign of quality.
The data behind the 2026 inspection surge
According to the EMA’s GMP/GDP Inspectors Working Group, the temporary extensions of certificate validity that were granted during the pandemic phased out at the end of 2024. As we move through 2026, NCAs have cleared the backlog and are back to a full schedule of on-site inspections.
Interestingly, recent industry data shows that over 22% of recent FDA warning letters cited issues specifically related to facility design and environmental control, proving that the Premises part of GMP is under more scrutiny than ever before.
The move toward the Quality Management System Regulation represents a move in global harmonization for our sector. According to a recent analysis by Hogan Lovells, the FDA is focusing on how these updated current good manufacturing practice requirements now align with the ISO 13485:2016 standard.
I expect that upcoming guidance will further clarify the specific documentation needed for premarket submissions, particularly around design controls and manufacturing processes.
This alignment makes it easier for teams to move through both U.S. and international regulations while determining how to obtain GMP certification for new devices. Consolidating these requirements shows that the agency helps manufacturers manage their compliance obligations with much greater efficiency.
Conclusion
Scilife takes the weight off your shoulders by automating your quality events and making sure your training records are always ready for an inspector to see.
Keeping all your data in one place allows you to remove manual processes that usually lead to those stressful audit findings. We are here to help you keep your facility running as a high-quality operation without the massive administrative headache. Then you can get back to what actually matters. Your products and your patients.
FAQs
How long does it take to get GMP certification?
The timeline usually ranges from 2 to 12 months. This greatly depends on a manufacturer's size, the current quality system maturity, and readiness for audits. This includes the time needed for a gap analysis, personnel training, and the scheduling of the official audit.
How long is a GMP certificate valid?
In the EU, a certificate is typically valid for three years, unless a follow-up inspection is triggered by a quality defect or a significant change to the facility.
How much does GMP certification cost?
Costs vary based on site size and complexity. You must factor in official agency fees, internal labor for preparation, and the cost of quality management software to maintain the system.
Is GMP certification mandatory?
Yes. If you manufacture or import medicines for the US or EU markets, compliance with GMP is a strict legal requirement.
Who will provide a GMP certificate?
In the EU, it is provided by your National Competent Authority. In the US, for domestic marketing purposes, it is often provided by an authorized third-party auditing firm.
.jpg)









