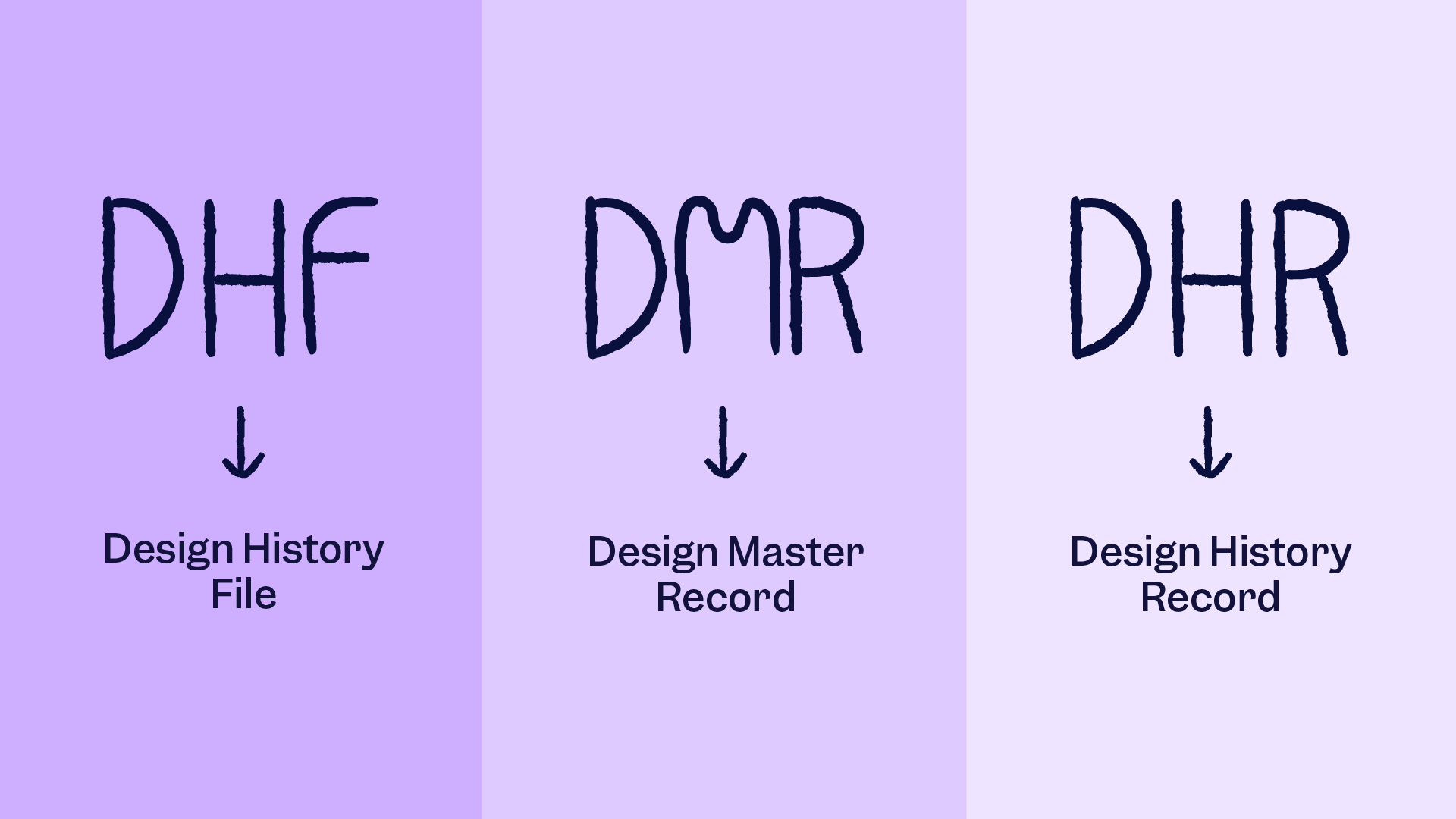
Unlike futuristic tech and devices, the medical device industry prefers simple designs. A simple to use, easy-to-use interface is crucial in devices providing life-saving treatment. However, that does not mean that designs do not take priority in medical devices. Device designs are just as important as their history.A design history file keeps a log of all initial information and changes made to the device design since its inception. It plays an important role in both safety and compliance.
In this article, we’ll dive into the FDA design history file requirements, the ISO 13485 design history file, how the DHF establishes the design control process, the common pitfalls, and how design history file software can help.
Let’s dive in!
Key takeaways
What is a design history file?
A design history file contains all the designs and updates of a medical device during its lifetime.
It contains all the necessary data to show that a medical device was properly designed and developed by following specific design control steps.
While the FDA enforces regulations to ensure quality, the DHF has a purely organizational advantage as well.
The design history files show that proper steps have been maintained throughout the process. This is beneficial because the details of the process stored in DHF also help with any future design changes and functional adaptations at your organization.
FDA design history file requirements
According to the Code of Federal Regulations Title 21 (FDA 21 CFR), Subchapter H – Medical Devices, Part 820 – Quality System Regulation, Subpart C – Design Controls, Section 820, the U.S. Food Drug Administration (FDA) states the following about a DHF:
(j) Design history file. Each manufacturer shall establish and maintain a DHF for each type of device. The DHF shall contain or reference the records necessary to demonstrate that the design was developed in accordance with the approved design plan and the requirements of this part.
ISO 13485 design history file requirements
Manufacturers in the US do not have only the FDA to consider, but also international standards such as ISO 13485:2016, which says that:
"The organization shall maintain a design and development file for each medical device type or medical device family. This file shall include or reference records generated to demonstrate conformity to the requirements for design and development and records for design and development changes."
From 2026, the DHF requirement is being replaced by the Quality Management System Regulation. However, the QMSR basically says the same thing as per ISO 7.3.10.
So, as of now, the official requirement of a collection of files similar to a design history file is still mandatory.
Here is a short summary of what the FDA design history file requirements entail:
- Each device with its individual design should have a DHF. However, it applies to devices with unique designs. If the design of two devices is the same, or same type, they can have a single DHF.
- The actual DHF may not have all the record details, but it does need to have the references to where the record is stored. However, for clarity, it’s better to have the records in print in the DHF. This goes for the QMSR too. If the records are referenced in DHF, they should be stored in QMS files. Needless to say, all files (referenced or otherwise) should be protected and stored properly.
- The DHF requires that the development process go according to the design plan AND design control. As such, it also reflects the compliance of the design and its suitability for product development.
Recommended learning: We know you probably have a lot of questions, so we’ve put together a complete guide to help you choose the right eQMS by asking the right questions.
How does the DHF establish the design control process?
FDA Section 820.30 requires that all manufacturers of Class II and Class III devices, as well as those of some Class I devices (1), establish and maintain procedures to control the medical device’s design, and (2) ensure that all requirements are met as specified in the design.
Hence, the DHF is a complete document showing a medical device’s concept, design, development, and testing. While it does serve a regulatory purpose, the DHF acts more as a lifetime record of a device design, ensuring its integrity.
Since the DHF captures the entirety of a device design and development, it begins with the very first design stage. As each phase of the design process advances, so does the file compilation of DHF.
The design control phases below show the steps of a complete medical device design and development. Associate documents of each step must be recorded in the DHF.

Design and development planning
This includes a plan for your design so that your medical device can be developed in accordance with it. This stage is about understanding your product, identifying the solution/treatment it’s trying to deliver, and planning the best way to deliver it. Flow charts and Gantt charts can prove helpful for this step.
Design input
This step includes established design inputs that address the intended use and user needs of the medical device. At this stage, the design concepts get more specific based on user requirements, state-of-the-art designs, safety protocols, potential market, etc.
Design outputs
Design outputs provide a device design that is compliant with the acceptance criteria that were defined during the design input step. The design outputs are tangible, specific designs like blueprints or prototypes. The device design history file will contain all details of this output.
Design review
The blueprint/design made in the design output step is reviewed in the next step. A design review is done systematically with several factors in mind, including efficiency of the device and safety.
After a device design passes the review process, a common practice is to “lock” the design and base the device on this version. Changes to the “locked” design can be made, but they need to go through a due review process. The DHF contains all the review items and outcomes.
Design verification
At the design verification stage, all designs go through a systematic review system in order to ensure that they align with the design input. After meticulous evaluation and tests, when a device design is passed as verified, the DHF documents the design validation process and the approved results.
Design validation
This step includes procedures for design validation and the results of your design validation. Design validation document shows that your design specification meets the user’s needs and requirements, for instance, a validation report.
Recommended learning: Design verifications VS validation - dive into our comparison article!
Design transfer
At the design transfer phase, the final design is manufactured, ensuring all design input components are maintained. The DHF contains documents for product specifications that are developed in compliance with this part and a description of the process used.
This step ensures that the established design is properly and effectively transferred into the product.
Design changes
Every DHF includes a part for design change in case of any changes needed relating to medical devices. Changes can be conducted using a simple change control request form or a change control management system.
Design history file
The DHF is one of the design controls; however, it also plays a role in maintaining records of all other design control parameters as a final step for the information generated by all other design controls.
[hand]
Design history file checklist: What belongs in your design history file?
No matter how simple the compilation of files seems, you are bound to miss a thing or two without a design history file checklist. Here is our simple, ready-to-use checklist for your use!
Planning
- Design and development plan (objectives, responsibilities, timeline)
- Risk management plan (per ISO 14971)
- Regulatory strategy (FDA, EU MDR, etc.)
Design Inputs
- User needs
- Design requirements (functional, performance, safety)
- Applicable standards (ISO, IEC, ASTM, etc.)
Design Outputs
- Engineering drawings & schematics
- Software architecture/Code documentation (if applicable)
- Bill of Materials (BOM)
- Manufacturing Instructions
Design Review
- Documented design reviews at key stages
- Review minutes & sign-offs
Design Verification
- Verification protocols (test plans)
- Verification reports (requirements met?)
Design Validation
- Clinical evaluation / Human factors studies
- Validation protocols & reports
- Usability studies
Design Transfer
- Manufacturing transfer documentation
- Supplier qualification records
- Device Master Record (DMR) references
Design Changes
- Change control records
- Rationale for each change
- Impact analysis
Design History Summary
- DHF Index or Table of Contents
- Confirmation that all required records are included
Common pitfalls when compiling the DHF
Tackling design control audits without any findings is not easy.
However, it depends on how you understand and apply the key steps. Organizations can make mistakes, and those mistakes do not come cheaply. Plus, it will be time-consuming to fix them.
Here are the most common pitfalls of a DHF:
Not having a DHF when designing
Your DHF should contain all documents that are created during the product development phase of your new medical device. But it is crucial to generate those documents while you are designing the product, not after.
Often, organizations rush to move the design control steps to get ready for mass production and enter the market as early as possible. That typically means they hope to compile the DHF later on, which can be due to a lack of understanding of what DHF outputs are required.
A disorganized DHF
This is as crucial as not having it at the right time. As you go through the design control steps, you will record the outputs of each design control step.
Even if you do everything else as required, if you do not gather and organize the outputs, you will lose track of them all. This can happen more often to organizations that still maintain a paper-based quality management system (QMS): Missing files, plans, and reports without a signature, overdue actions – they all can lead to a possible finding if one of them is disclosed during an audit or inspection.
A disorganized document management system can make it difficult to control, track, maintain, or manage changes and revisions.
On the other hand, an eQMS that comes with a built-in document control solution can centralize and categorize all your company documents, so you can stop worrying about what’s where, what needs to be updated or approved, and who has access to what.
Including unnecessary documents
Making a bigger file than needed is exactly that – unnecessary. When starting, you may think that including as many files as possible in the DHF is a good idea.
But keep in mind that you will keep DHF throughout the lifecycle of the product. Therefore, maintaining a file full of unnecessary documents will become more and more difficult, while also increasing the risk of being questioned by auditors.
Auditors can take a bloated DHF as a sign of you not having enough knowledge or not having fully understood the idea of a DHF.
Authorization for compiling and maintaining the DHF
If you don’t have a digital document management system, you are likely to face problems when compiling and maintaining the DHF.
Unmanaged documents – such as uncontrolled, outdated e-mail printouts, or a shared folder with read-and-write access – are bad signs of an ineffective document management system.
This can lead to exponentially bigger problems and critical findings, rendering your DHF non-compliant.
No traceability to outputs
If outputs of the design control elements are not well documented, you do not have a proper DHF. The following step would be impossible, which is creating your DMR!

How design history file software can help keep your DHF organized and audit-ready
Creating a single, simple device’s DHF is easy enough. However, when you throw in multiple devices with unique, complex designs and tests, the DHF can become a huge regulatory headache with design history file software.
Not to mention, extracting specific files from the DHF when there is a design issue is time-consuming. Having a document management module, preferably integrated as part of your eQMS, works as your design history file software and can make things significantly easier and smoother. This is thanks to key capabilities such as:
Centralized control: Keep all DHF documents organized and accessible in one secure place.
Traceability links: Easily connect requirements, designs, tests, and changes across devices.
Version control and audit trails: Maintain compliance effortlessly with automatic updates and full history logs.
Compliant electronic signatures: Capture approvals in line with FDA 21 CFR Part 11 and EU Annex 11 requirements.
Quick search and retrieval: Find the exact file you need in seconds, not hours.
Collaboration-ready access: Enable cross-functional teams to work seamlessly without losing oversight.
Scilife Tip:
A well-maintained DHF is not just a regulatory requirement; it’s a strategic asset that safeguards compliance, supports quality, and simplifies audits. Using digital tools like design history file software makes managing it far easier and more reliable.
Conclusion: Make DHF easy with an eQMS
A Design History File is the backbone of medical device design compliance and a vital safeguard for patient safety.
By compiling and organizing your DHF alongside development, you not only meet FDA and ISO 13485 design history file requirements but also build a reliable foundation for future improvements, audits, and market success.
Avoiding the common pitfalls like late compilation, poor traceability, or bloated documentation can save you costly setbacks that you’ll be glad we told you about. And with the right digital tools like Scilife’s design and development solution, keeping your DHF structured, complete, and audit-ready becomes far simpler.